Accreditation Software
AQS provides software for both laboratories and accrediting bodies—aligning quality management and accreditation workflows around a shared goal: ensuring laboratory data is scientifically and legally defensible.
Most accreditation programs rely on disconnected tools—spreadsheets, email, and separate tracking systems for laboratories and accrediting bodies. This often leads to duplication, inconsistency, and unnecessary risk.
AQS brings both sides of the process into alignment with two complementary applications.
AB Manager & QA Manager
Two systems. One goal: defensible laboratory data.
Purpose-built for laboratories and accrediting bodies, these systems reflect the same workflows, terminology, and expectations—supporting the full lifecycle of laboratory quality and accreditation.
AB Manager
Accreditation Management for Accrediting Bodies
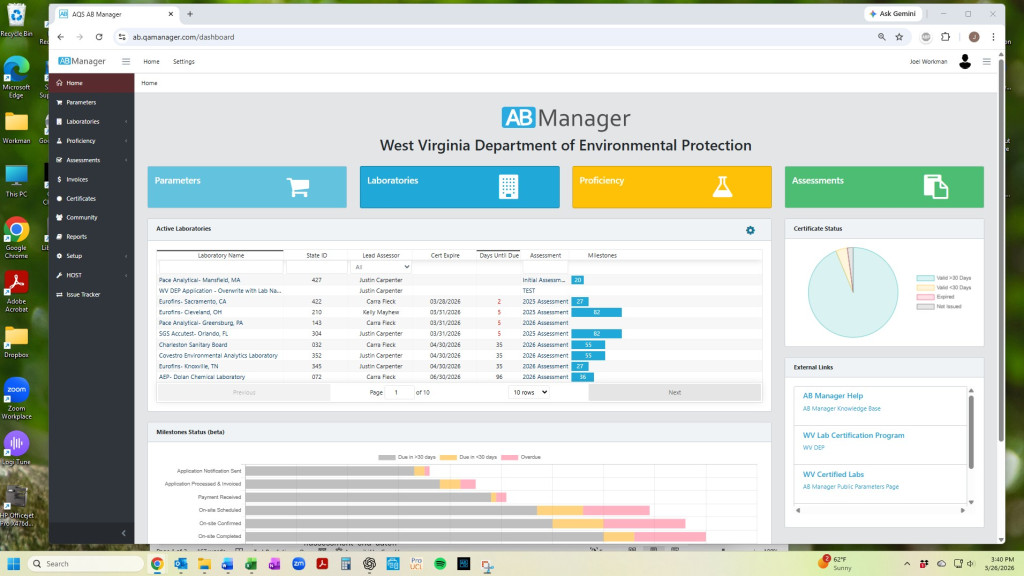
AB Manager is designed specifically for environmental laboratory accrediting bodies to manage the full accreditation lifecycle—from application through assessment, reporting, and certificate issuance.
Key capabilities:
- Application and reapplication tracking
- Accreditation scopes and parameter management
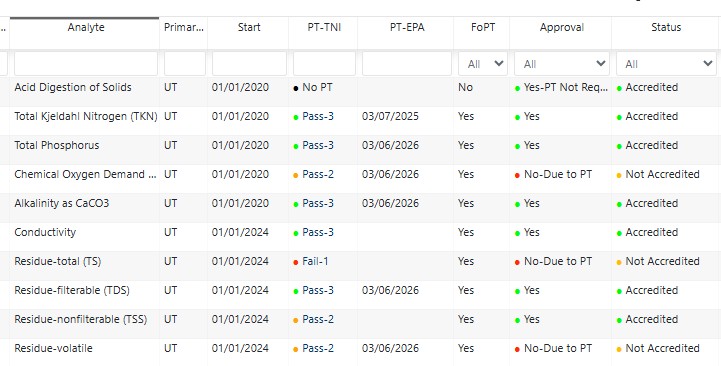
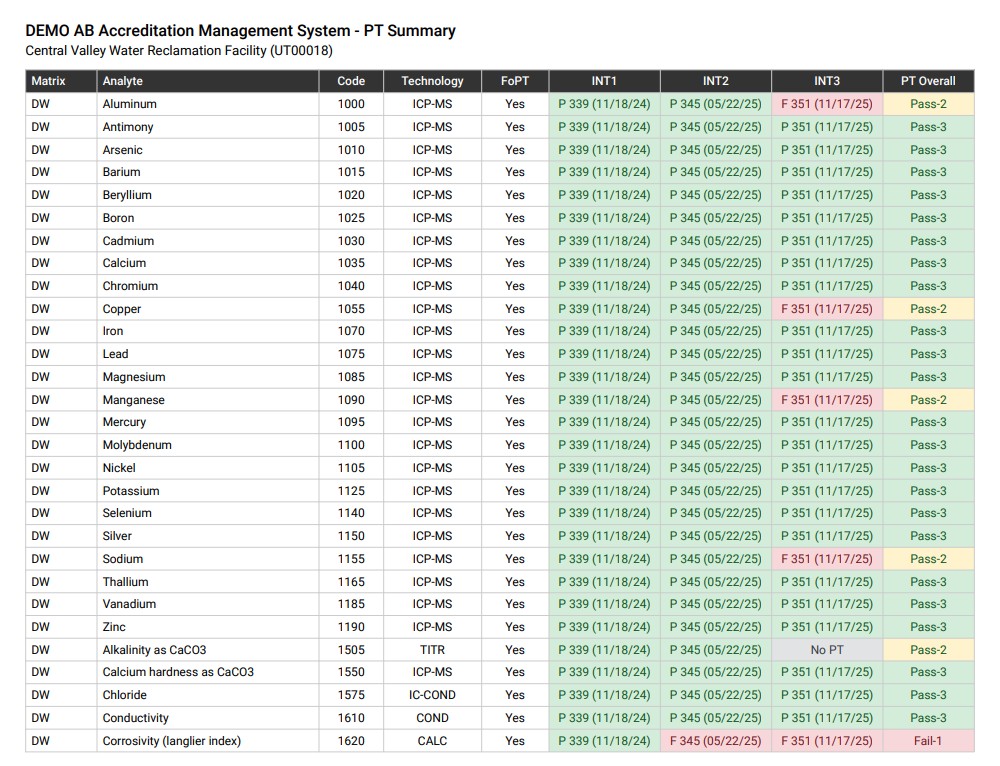
- Proficiency testing (PT) evaluation
- Assessor scheduling and coordination
- Onsite assessments and inspection checklists
- Findings, responses, and corrective action tracking
- Certificate generation and reporting
AB Manager helps accrediting bodies operate consistently, improve defensibility, and reduce administrative burden.
QA Manager
Quality Management for Laboratories
QA Manager is a structured quality management system designed to help laboratories stay organized, compliant, and audit-ready.
Built for laboratory directors, quality managers, and technical staff, QA Manager supports the core elements of a laboratory quality system.
Key capabilities:
- Document control and version management
- Training tracking and personnel records
- Recurring tasks and scheduled activities
- Internal audits and inspections
- Corrective and preventive actions (CAPA)
- Centralized tracking of quality system activities
QA Manager helps laboratories maintain control of their quality systems and align with accreditation requirements.
Learn More…
Benefits of a Complementary Approach
AB Manager and QA Manager are designed to work together—supporting different roles within the same overall accreditation process.
- Laboratories use QA Manager to implement and maintain quality systems
- Accrediting bodies use AB Manager to review, verify, and coordinate those systems
Together, they represent both sides of accreditation:
- what laboratories do to maintain quality
- what accrediting bodies evaluate to confirm it
Two systems. One goal: defensible laboratory data.
What This Provides
- Alignment with TNI standards and accreditation requirements
- Consistent checklist structures and evaluation logic
- Reduced duplication between laboratories and accrediting bodies
- Improved communication and transparency
- More defensible and traceable processes
Whether used independently or together, both applications support a more structured, consistent, and reliable approach to laboratory quality and accreditation.